Research Overview
Our research is motivated by the high mortality associated with cardiovascular disease, particularly the risk of death following myocardial infarction. Although current therapies have improved acute survival, they do not adequately repair damaged heart tissue or prevent long-term functional decline. The overarching goal of our research is to advance fundamental knowledge and develop effective therapeutic strategies for heart disease. Our lab is dedicated to creating innovative bioengineering and regenerative approaches that promote cardiac repair, restore function, and improve patients’ quality of life.
Our current research focuses on three main areas: (1) advancing cardiac cell therapy by harnessing the regenerative potential of human stem cells and developing effective cell delivery strategies; (2) promoting angiogenesis both in vitro, to generate vascularized bioscaffolds for tissue engineering applications, and in vivo, to rescue ischemic tissues; and (3) designing innovative microfluidic chip platforms to study heart development, disease mechanisms, and therapeutic responses.
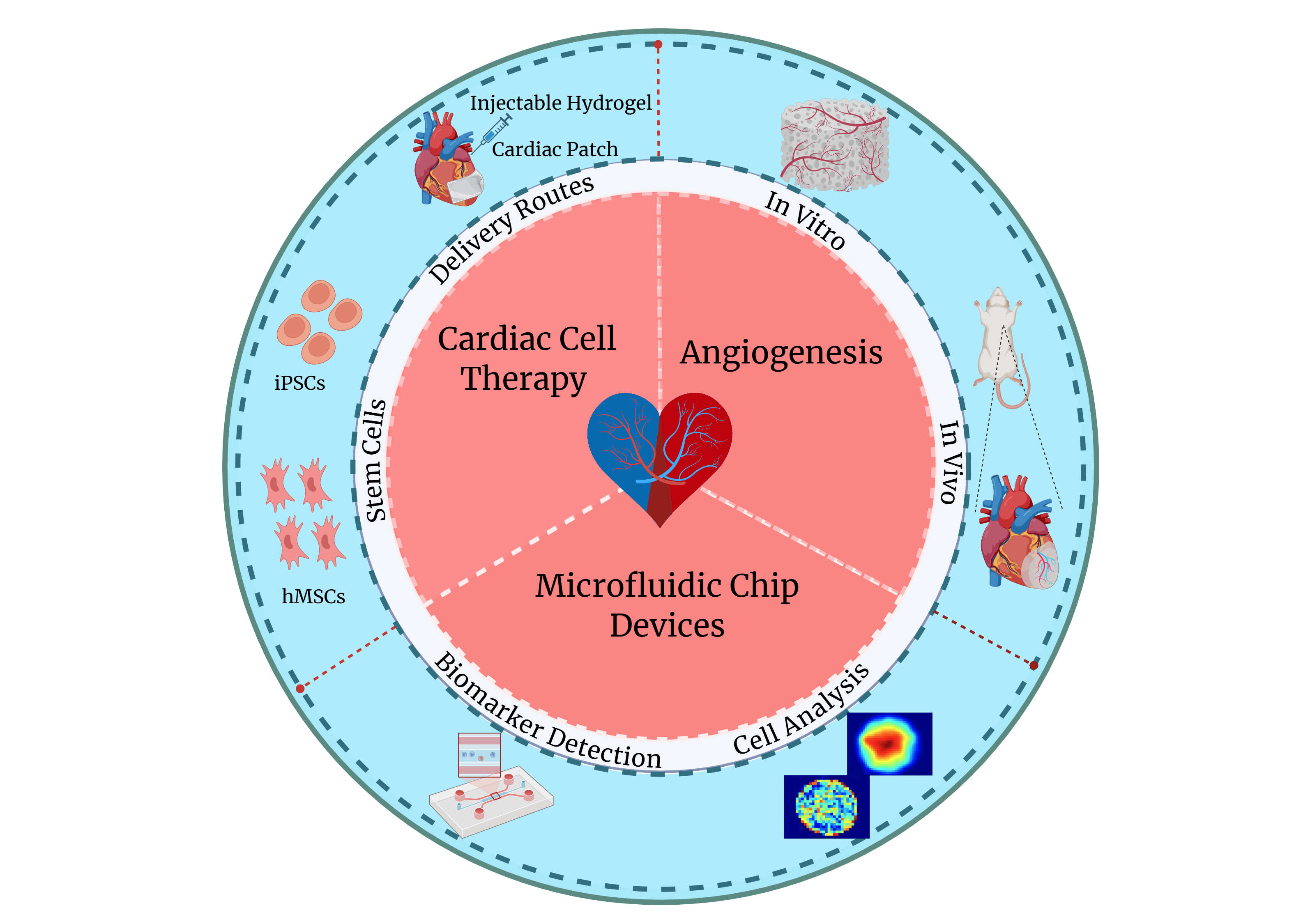
Advancing Cardiac Cell Therapy
Stem cell–based cardiac therapy holds great promise for the treatment of myocardial infarction; however, its therapeutic benefits have often been limited and transient. Key challenges include poor cell survival and engraftment after transplantation, the hostile microenvironment of the injured myocardium, and adverse effects from immune responses triggered by cardiac injury.
To address these challenges, our lab has established a collaborative research program that integrates diverse bioengineering tools and technologies, including decellularized extracellular matrix scaffolds, cell sheet engineering, and injectable hydrogel systems. Through these approaches, we aim to enhance cell retention, survival, and functional integration, ultimately improving the therapeutic efficacy of cardiac cell therapy.
Relevant publications from our lab:
- R. Shaik, J. Brown, J. Xu, R. Lamichhane, Y. Wang, Y Hong, G. Zhang. “Cardiac Matrix-Derived Granular Hydrogel Enhances Cell Function in 3D Culture” ACS Applied Materials & Interfaces, 2024 16 (43), 58346-58356
- M. Shah, P. KC, G. Zhang, “In Vivo Assessment of Decellularized Porcine Myocardial Slice as an Acellular Cardiac Patch” ACS Applied Materials and Interfaces, 2019, 11(27), 23893-23900.
- N. Patel, G. Zhang, “Stacked stem cell sheets enhance cell-matrix interactions” Organogenesis, 2014,10 (2), 1-7.
Promoting In Vitro and In Vivo Angiogenesis
Adequate vascularization is essential for effective tissue regeneration and functional recovery following ischemic injury. Insufficient angiogenesis limits oxygen and nutrient delivery, contributing to cell death and poor tissue integration in both engineered constructs and damaged native tissues. Promoting robust and functional blood vessel formation therefore represents a critical challenge in cardiovascular tissue engineering and regenerative medicine.
Our lab focuses on developing bioengineering strategies to stimulate angiogenesis both in vitro and in vivo. In vitro, we engineer vascularized bioscaffolds and 3D tissue constructs by integrating endothelial cells, stromal cells, and pro-angiogenic cues to support stable microvascular network formation. In vivo, we design biomaterial- and cell-based approaches to enhance neovascularization in ischemic tissues, aiming to restore perfusion, improve tissue survival, and support long-term functional recovery. Together, these efforts seek to bridge fundamental angiogenesis biology with translational strategies for cardiac and ischemic tissue repair.
Relevant publications from our lab:
- ·R. Shaik, J. Xu, Y. Wang, Y. Hong*, G. Zhang*, “Fibrin-Enriched Cardiac Extracellular Matrix Hydrogel Promotes In Vitro Angiogenesis”, ACS Biomaterials Science & Engineering, 2023, 9, 877-888.
- KC, M. Shah, J. Liao, G. Zhang, “Prevascularization of Decellularized Porcine Myocardium Slice for Cardiac Tissue Engineering” ACS Applied Materials and Interfaces, 2017; 9 (3), 2196-2204.
- M.E. Jeffords, J. Wu, M. Shah, Y. Hong, G. Zhang, “Tailoring Material Properties of Cardiac Matrix Hydrogels to Induce Endothelial Differentiation of Human Mesenchymal Stem Cells” ACS Applied Materials & Interfaces, 2015, 7 (20), 11053–11061.
Designing Microfluidic Platforms for Cardiac Research
Microfluidic devices provide powerful platforms for detecting disease-related biomarkers and probing cellular behavior at the single-cell level. Over the past decade, we have created microfluidic platforms for a range of applications, including highly sensitive, rapid, and low-volume analysis of biomarkers, stem cell secretome analysis, single-cell electrical stimulation and mapping.
Building on this foundation, our ongoing research focuses on developing next-generation microfluidic chip platforms that integrate human cardiac cells, biomaterials, and tightly controlled biochemical and mechanical stimuli to generate physiologically relevant heart-on-chip models. By combining microengineering with stem cell and tissue engineering technologies, our microfluidic platforms serve as powerful research tools for mechanistic studies and therapeutic development in cardiovascular disease.
Relevant publications from our lab:
- L.Ouyang, H. Chen. R. Xu,R. Shaik, G. Zhang, J. Zhe. “Rapid Surface Charge Mapping Based on a Liquid Crystal Microchip”, Biosensors, 2024, 12,199
- L. Ni, P. KC, E. Mulvany, G. Zhang, J. Zhe, “A microfluidic device for noninvasive cell electrical stimulation and extracellular field potential analysis” Biomed Microdevices, 2019;21(1): 20.
- P. KC, F Liu, J. Zhe, G. Zhang, “Development and Comparison of Two Immuno-disaggregation Based Bioassays for Cell Secretome Analysis” Theranostics, 2018, 8(2), 328-340.