Harvesting gas vesicles from blue-green algae for medical and industrial applications
Gas vesicles, which are the components of gas vacuoles, provide buoyancy to cyanobacteria allowing them to position in a place where they take full advantage of the surrounding light and nutrient conditions. These vesicles are effective gas microcarriers with potential industrial and medical applications. Unfortunately, they are too delicate to be collected by common methods such as filtration and centrifugation. In this study, we are investigating alternative methods of harvesting and collecting the gas vesicles.
For gentle cell harvesting, we have examined the natural flotation of the buoyant cells under various light intensities and cell concentrations. The flotation efficiency is highest under dark conditions with high cell concentrations. The cells collected by flotation are then broken by osmotic shock in a 0.7 M sucrose solution. The released gas vesicles can be collected by repeated gentle centrifugation. This process is rather lengthy and laborious. We are developing a more effective collection method by inducing selective flocculation, so that the flocculated gas vesicles float more rapidly while the cell debris sinks.
A method to crosslink the collected gas vesicles for improved stability against moderate heat and denaturing surfactants has been developed. The gas vesicles can be chemically sterilized. Good oxygen permeability through the gas vesicles has been measured. The use of gas vesicles as gas microcarriers to enhance oxygen supply in cell culture has also been demonstrated. We are developing other potential medical applications of the gas vesicles.
 |
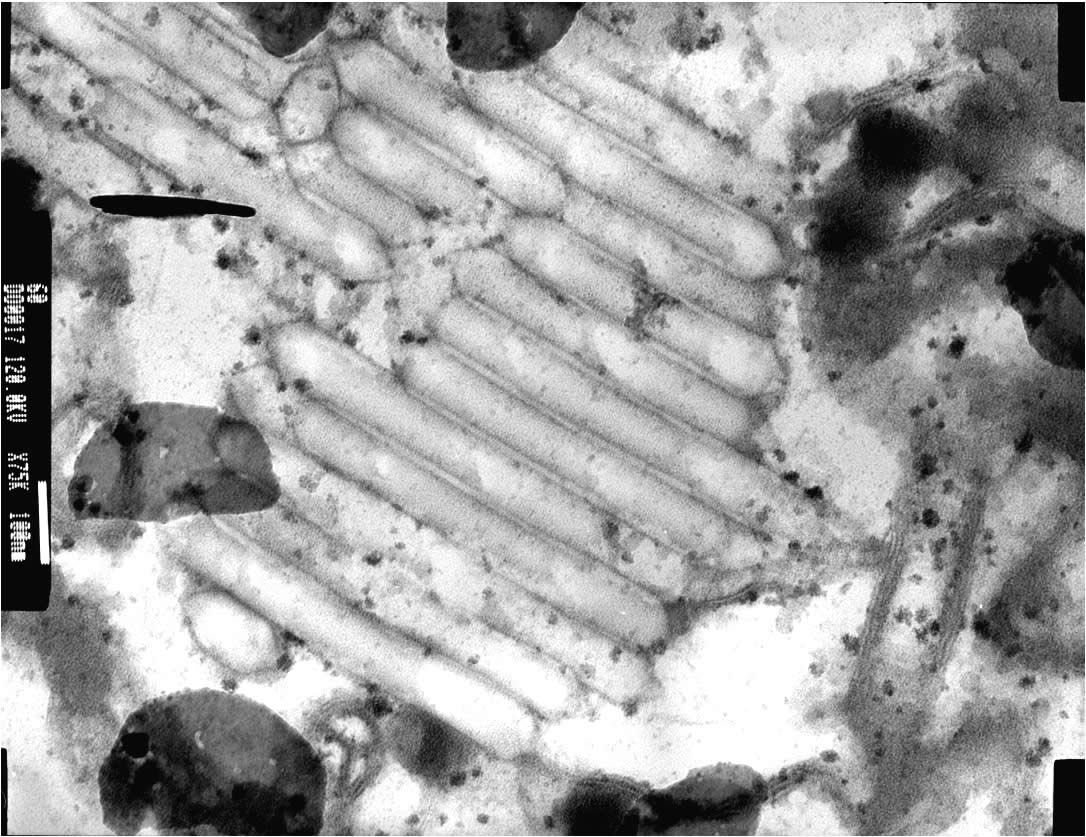 |
|
| Vegetative (photosynthetic cells) and heterocysts cells of Anabaena flos-aquae. Arrows point to heterocysts | SEM image of gas vesicles in A. flos-aquae |
Publications:
Use of cyanobacterial gas vesicles as oxygen carriers in cell culture. (A. Sundararajan and L.-K. Ju) Cytotechnology 52(2), 139-149 (2006). DOI Link
Effects of polyethyleneimine on cyanobacterium Anabaena flos-aquae during cell flocculation and flotation. (S. A. Arrington, M. J. Zeleznik, D. W. Ott and L.-K. Ju) Enzyme and Microbial Technology 32 (2), 290-293 (2003).
Polyethyleneimine-induced flocculation and flotation of cyanobacterium Anabaena flos-aquae for gas vesicle production. (M. J. Zeleznik, J. M. Segatta, and L.-K. Ju) Enzyme and Microbial Technology 31, 949-953 (2002). DOI Link
Evaluation of oxygen permeability of gas vesicles from cyanobacterium Anabaena flos-aquae. (A. Sundararajan and L.-K. Ju) Journal of Biotechnology 77, 151-156 (2000). DOI Link
Glutaraldehyde treatment of proteinaceous gas vesicles from cyanobacterium Anabaena flos-aquae. (A. Sundararajan and L.-K. Ju) Biotechnology Progress 16, 1124-1128 (2000). DOI Link
Flotation characteristics of cyanobacterium Anabaena flos-aquae for gas vesicle production. (S. Kashyap, A. Sundararajan and L.-K. Ju) Biotechnology and Bioengineering 60, 636-641 (1998). DOI Link